Introduction
As China’s medical system aligns with international standards and innovative drug R&D accelerates globally, an increasing number of expatriates are choosing China as a destination for participating in new drug clinical trials. Having joined the ICH (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use), China’s clinical trial standards are highly consistent with those in Europe and the US. It offers unique advantages such as high approval efficiency, centralized top-tier hospital resources, free treatment costs, and subsidies.
It must be made clear: Not all trials accept foreign participants. Pharmaceutical companies often set nationality or ethnic restrictions due to ethnic differences, drug metabolism characteristics, and registration requirements. This guide is designed for expatriates or visitors in China, systematically explaining eligibility, search channels, the full process, and rights protection to help you participate safely and compliantly.
1. Core Advantages
- Cutting-edge Therapies: Get priority access to innovative drugs not yet on the market, especially suitable for patients with chronic diseases, cancer, and rare diseases.
- Completely Free: All trial-related drugs, examinations, and treatments are free. Some projects provide transportation and nutrition subsidies.
- Top-tier Medical Care: Managed by authoritative expert teams from top-tier hospitals with intensive follow-up and meticulous monitoring.
- International Standards: Strictly follows GCP and ICH norms; ethical review and data management are standardized.
- No Queuing: Green channel in International Departments/Special Needs Clinics, equipped with translation support for a smooth process.

2. Regulatory and Policy Framework
- Core Regulations: “The Drug Administration Law of the People’s Republic of China”, “Regulations for Drug Registration” (2022 Revision), “Good Clinical Practice” (GCP).
- Registration Requirements: All clinical trials must be registered with the Chinese Clinical Trial Registry (ChiCTR) and approved by the Ethics Committee before starting.
- Data Mutual Recognition: The NMPA recognizes qualified overseas clinical data; international cooperation involving human genetic resources must be filed according to regulations.

3. Eligibility and Restrictions (Hard Thresholds)
| Restriction Dimension | Description | Remarks |
|---|---|---|
| Ethnicity/Race | Core Threshold. Local registration trials often require “Chinese citizens only” to obtain specific population data; MRCTs usually accept foreigners. | Drug metabolism (PK/PD) has ethnic differences; read protocols carefully. |
| Visa/Residency | Must hold a long-term residence permit (Work, Reunion, Private Affairs). | Tourist Visas usually do not qualify as they cannot guarantee long-term follow-up. |
| Language Ability | Must understand the Chinese or English version of the “Informed Consent Form” (ICF). | Large top-tier hospitals usually provide English support or allow personal translators. |
4. Advantageous Disease Fit (Higher Success Rate)
Combined with China’s advantageous fields in clinical trials, the following directions have higher openness to foreigners:
- Oncology: Liver cancer, Esophageal cancer, Nasopharyngeal cancer (More MRCTs for cancers with high incidence in China).
- Rare Diseases: Supported by national policy; ethnic restrictions are usually relaxed, focusing on biological indicators.
- Metabolic/Chronic Diseases: MRCTs for innovative drugs targeting specific targets.
5. How to Find Participable Clinical Trials (Channel Analysis)
Finding clinical trials is essentially a balance between “Information Breadth” and “Connection Efficiency”. Here is a deep comparison of three mainstream channels:
5.1. Official Registration Platforms (Shotgun Approach)
- Platforms: Chinese Clinical Trial Registry (ChiCTR)
- Operation: Input disease keywords, filter for “Recruiting” projects.
- Expert Comment: This is the most basic “Yellow Pages,” suitable for background checks. However, the phone numbers listed are often office landlines. For foreigners, language barriers plus long administrative processes may cause you to miss the best treatment window.
5.2. Hospital International Departments (Precision Strike Mode)
- Targets: Top hospitals like Peking Union Medical College Hospital, Shanghai Ruijin, Guangzhou Zhongshan Tumor, etc.
- Operation: Register at the International Department and ask the doctor directly: “Are there any MRCTs for my condition?”
- Expert Comment: This is the path with the highest success rate. International department doctors are usually more aware of global R&D trends and have the authority to recommend patients directly. The downside is you have to ask one by one, and if the hospital has no relevant projects, you make a wasted trip.
5.3. Third-party Assistance (Efficient Shortcut Mode) like MedChinaGuide.com
- Operation: Professional agencies conduct medical record pre-evaluation and channel docking.
- Expert Comment: For foreigners unfamiliar with the Chinese medical environment, this is the most efficient way to avoid the “Ethnic Restriction” minefield and quickly find “Expats Welcome” projects.

6. Real Case References
Why look at cases? Real successful enrollment experiences help you judge: Is there a chance for your condition? How hard is it for foreigners to join?
Case 1: Rare Disease/Gene Therapy (High Difficulty Enrollment)
- Patient Background: 11 years old, Guinean, suffering from severe Sickle Cell Anemia.
- Difficulty: Rare blood type (Panda blood), difficult local transfusions, ineffective conventional drugs.
- Solution: Through the gene editing clinical trial (BRL-102) collaborated by Bioray Biotech and Guangxi Medical University.
- Result: Successfully enrolled, freed from transfusion dependence after treatment, hemoglobin returned to normal.
- Insight: Even for rare diseases, as long as China has cutting-edge gene editing/cell therapy projects, ethnic restrictions are often looser because pharmaceutical companies urgently need diverse clinical data.
Case 2: Relapsed Tumor/CAR-T Therapy (Efficient Enrollment)
- Patient Background: 45 years old, Foreigner, Relapsed/Refractory Multiple Myeloma.
- Difficulty: No drugs available in home country, waiting time too long.
- Solution: Went directly to Beijing United Family Hospital, docked with the CAR-T cell immunotherapy project.
- Result: Completed evaluation and enrollment within 2 weeks, treatment cost was only 1/5 of that in Europe/US.
- Insight: For hematological tumors, China has many CAR-T projects and fast approval; contacting the International Department directly is the most efficient path.
Case 3: Solid Tumor/Clinical Trial (Active Pursuit)
- Patient Background: 54 years old, Polish doctor, Advanced gastric cancer with lung metastasis.
- Difficulty: Standard treatment failed, told no cure by conventional channels.
- Solution: Family actively emailed the Cancer Hospital, Chinese Academy of Medical Sciences, applying for the TILs (Tumor Infiltrating Lymphocytes) clinical trial.
- Result: The hospital was moved by their sincerity, allowed enrollment after evaluation, and the family was even willing to move to China for this.
- Insight: For solid tumors, if official channels don’t work, professional agencies or directly contacting experts (e.g., email self-recommendation) can sometimes create miracles.
7. Step-by-Step Participation Process
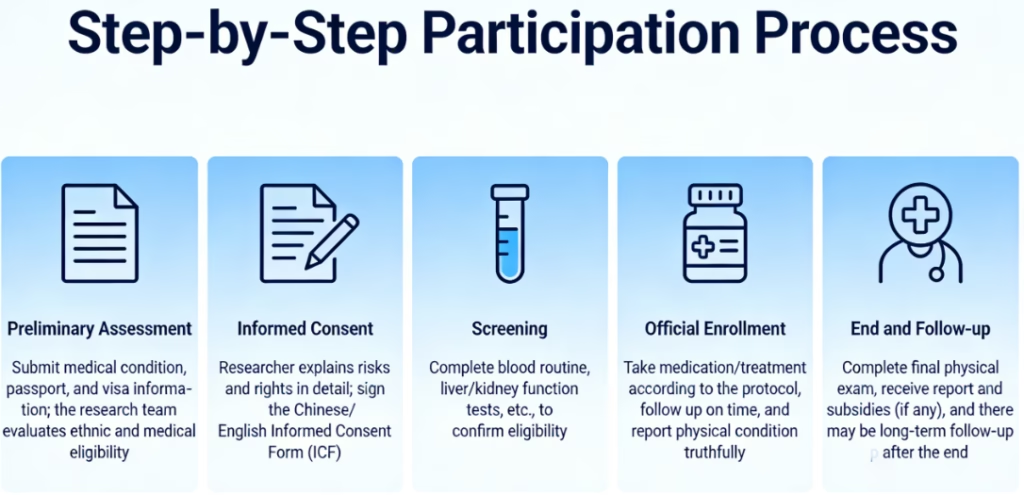
- Preliminary Assessment: Submit medical condition, passport, and visa information; the research team evaluates ethnic and medical eligibility.
- Informed Consent: Researcher explains risks and rights in detail; sign the Chinese/English Informed Consent Form (ICF).
- Screening: Complete blood routine, liver/kidney function tests, etc., to confirm eligibility.
- Official Enrollment: Take medication/treatment according to the protocol, follow up on time, and report physical condition truthfully.
- End and Follow-up: Complete final physical exam, receive report and subsidies (if any), and there may be long-term follow-up after the end.
8. Key Considerations
- Ethnic Difference Risks: Foreigners may experience side effects not observed in Chinese people (race-specific risks); close monitoring is required.
- Rights Protection: Enjoy the same rights as Chinese subjects; free treatment and compensation for trial-related injuries are available.
- Prohibitions: Do not conceal medical history or concomitant medication; do not leave the country without permission (otherwise deemed withdrawal); can withdraw unconditionally at any time.
9. FAQ
- Q: Do I need to pay?
- A: No. Trial drugs, examinations, and treatment costs are fully covered; some projects provide subsidies.
- Q: Can I participate with a tourist visa?
- A: Usually not. Trial cycles are long; long-term residence visas are required to ensure follow-up compliance.
- Q: How to confirm if a trial is compliant?
- A: Check the ChiCTR official website to confirm the project is registered and the ethics approval is publicized.
10. MedChinaGuide.com Professional Services
We are dedicated to helping expatriates in China participate in clinical trials safely and efficiently:
- Precision Matching: Filter for compliant trials accepting foreigners, avoiding ethnic restriction projects.
- Full-process Escort: Provide medical record translation, eligibility pre-evaluation, hospital docking, and follow-up reminders.
- Cross-border Support: Assist with special processes involving international multi-center trials.
“The team helped me find a suitable trial, arranged translation, and walked me through every step. I got access to new treatment I couldn’t get back home.”
— Expat patient, 2026
Data Sources: NMPA, ChiCTR, ICH-GCP, Hospital Official Websites, Medchinaguide search
Last Updated: 2026-04-01


